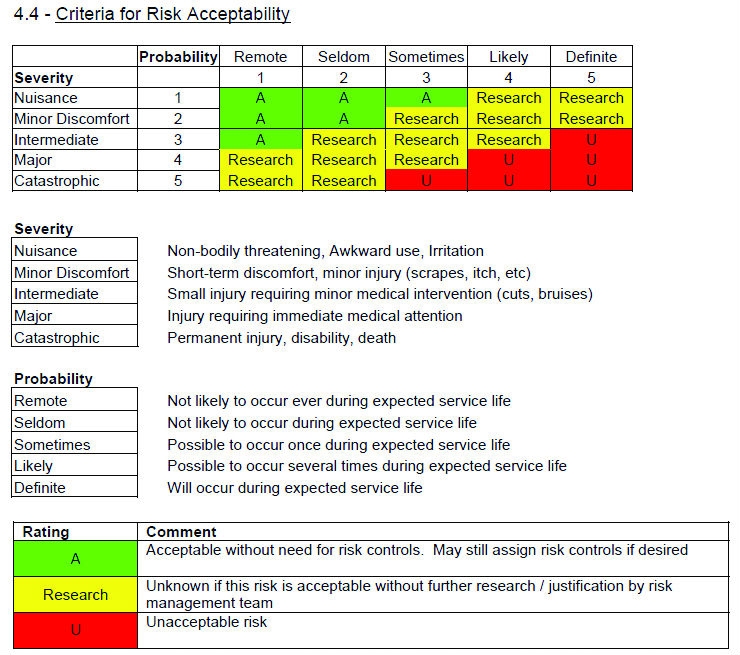
If any risk is judged unacceptable, it should be reduced to acceptable levels by appropriate means. Manufacturers are expected to identify possible hazards associated with the design in both normal and fault conditions. Risk analysis, or hazard analysis, is a structured tool for the evaluation of potential problems which could be encountered in connection with the use of taking a drug, or using a medical device. Risk Analysis plays a key role in the development of medical devices design. Risk management involves the identification, understand, control, and prevent failures that can result in hazards when people use medical devices. This implies that the device should be safe and effective. Researchers in charge to develop new medical devices are faced with the complex task of making a medical device safe for human use. 
Moreover, these devices can be used for daily patient care as well as for medical scientific purposes. 
They may either be an instrument, an apparatus or a material. Medical devices developed for human application are used for diagnostic or treatment purposes.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
